|
The scientists published their explosive results last week in the journal Nature Chemistry. It also may explain why the explosions can be finicky, and vary depending on the state of the metal and any contaminants present. Thats why potassium gives a more violent reaction with. This reduces the ionic mobility which in turn reduces the speed of the molten metal. Also, the hydrated radius of lithium is the greatest out of all alkali metals. The molten metal spreads over the water and exposes a larger surface to water. It will give off hydrogen and eventually catch fire. The heat of reaction is sufficient to melt it. However, chemically, potassium reacts with water violently. So, this is an example of an Exothermic reaction. The authors.suggest is necessary for all of the explosive reactions between water and the elements in the first column of the periodic table. Potassium is less dense than water, so it can float on water. Potassium reacts rapidly and intensely with water, forming a colourless basic potassium hydroxide solution and hydrogen gas, according to the following reaction mechanism: 2K (s) + 2H 2 O (l) -> 2KOH (aq) + H 2 (g) This is an exothermal reaction and potassium is heated to such an extend that it burns a purple flame. When Potassium reacted with water it produces potassium hydroxide and hydrogen with a large amount of heat. (Potassium compounds are also important to a lesser extent in the manufacture of explosives. 
Thus, charge repulsion causes the spikes. Table of Contents Potassium - Compounds, Reactions, Elements: Of commercially produced potassium compounds, almost 95 percent of them are used in agriculture as fertilizer. Computerized simulations of this reaction showed the surface of the metal rapidly forms a large positive charge, and this charge repulsion leads to a rapid expansion and disintegration of the surface. The authors focused on what happens after the electrons leave: the metal that remains is a collection of charged ions.  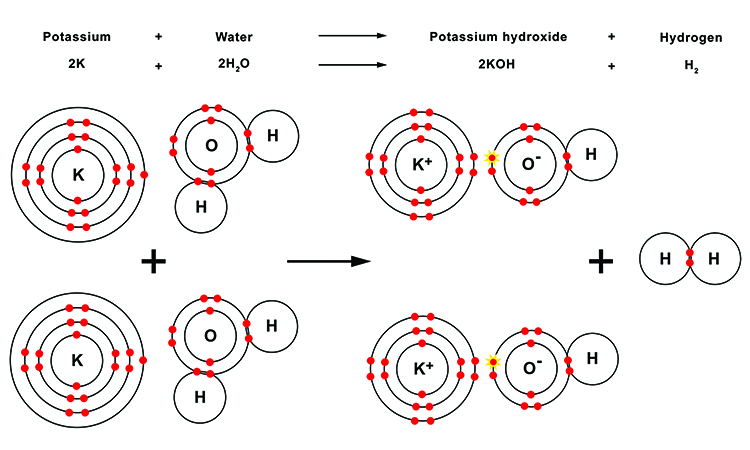
Timmer more fully explains what causes the spikes: (They concocted this exact mixture so that it could be a uniform sphere more complex shapes make the experiment impossible to closely observe and repeat.) Turns out, the metal shoots out into countless tiny spikes just after it hits the water, but this so-called "Coulomb explosion" happens extremely quickly, appearing visually for only less than a millisecond, at 0:47 in the video above. Scientists recently created an ultra-high-speed video of a drop of liquid sodium and potassium landing in water, filming from above and below to determine exactly what is going on.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
